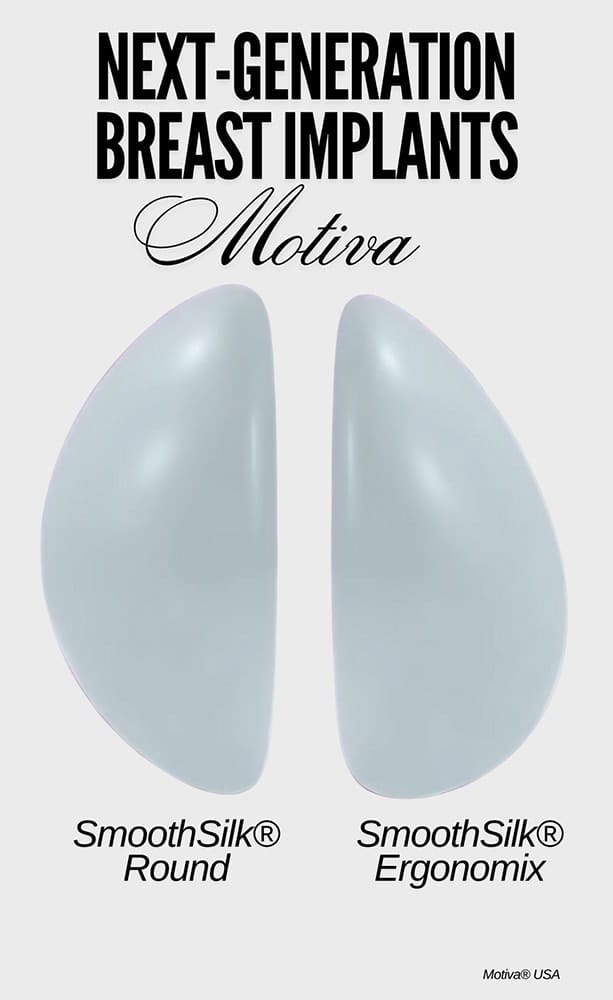
Motiva® 6th-generation breast implants became FDA-approved and commercially available in the USA in October 2024. Because Motiva® breast implants were first introduced to the market in 2010 and are used in dozens of countries around the world, they have been well-studied and documented in scientific journals, including “Nature”, the world’s leading scientific journal.
Why Choose Motiva® Breast Implants?
Motiva® breast implants stand out due to their unparalleled, exceptional safety features, advanced technology, innovative design, and unique features, focusing on achieving the feel, movement, and appearance similar to natural breast tissue. The reasons why so many individuals choose Motiva® breast implants include:
- The Natural Look and Feel of the Implant:
- Motiva® breast implants are known for their soft, natural feel and are stretchable and compressible, enabling them to closely mimic the look, feel, and movement of natural breast tissue.
- The unique ProgressiveGel® is a 6th-generation, highly cohesive, viscoelastic silicone gel that provides a natural, soft feel and cohesive shape retention, resulting in fewer folds, less friction, and an unprecedentedly low rupture rate.
- The revolutionary SmoothSilk® 4-micron, nano-textured surface of the outer shell that encases the gel filling is so unique that it was given its own category by the FDA: “4-micron”, behaving like a traditional smooth surface.
- The unique TrueMonobloc® shell, created by next-generation gel-bonding technology, unifies the implant’s shell, gel, and patch into one single, cohesive unit. By integrating all components of the implant into a single structure, implant flexibility and strength are enhanced, enabling natural movement, improved durability with a less than 1% rupture rate, and easier implant placement.
- The TrueMonobloc® shell with the SmoothSilk® 4-micron surface and advanced ProgressiveGel® silicone-gel filling forms one cohesive unit, making the implant feel like one entity, unlike traditional breast implants. The difference between the silky-feeling Motiva® implant and a traditional breast implant is immediately recognizable. Together, Motiva®’s technological features create a more natural contour, ensuring that the patient’s results feel as good as they look.
- The Body’s Reaction to the Implant:
- The revolutionary SmoothSilk® 4-micron, nano-textured surface – so unique that it was given its own category by the FDA – is designed for enhanced biocompatibility and optimized immune response of the body, improving tissue integration and promoting a low inflammatory response, low bacterial attachment, and low friction. These factors consequently produce a softer capsule (the collagen-based scar tissue that forms around every breast implant as a normal part of the body’s healing process) and greatly reduce the risk of capsular contracture to an impressively low 0.5% rate on average (capsular contracture being when the scar tissue that naturally forms around the implant tightens and becomes unusually hard).
- Together, they create breast implants that provide a gentle interface with surrounding tissue, feel soft and natural, adapt well to body movements, support a safer healing process, and prioritize long-term comfort and safety.
- The unique BluSeal® visual indicator of the barrier layer confirms that the implant shell is intact before surgery and that the integrity of the internal and external implant components is assured, ensuring quality assurance and patient safety.
- An optional RFID micro-transponder can be embedded in the implant, a technological innovation enabling non-invasive verification of the implant.
- The revolutionary SmoothSilk® 4-micron, nano-textured surface – so unique that it was given its own category by the FDA – is designed for enhanced biocompatibility and optimized immune response of the body, improving tissue integration and promoting a low inflammatory response, low bacterial attachment, and low friction. These factors consequently produce a softer capsule (the collagen-based scar tissue that forms around every breast implant as a normal part of the body’s healing process) and greatly reduce the risk of capsular contracture to an impressively low 0.5% rate on average (capsular contracture being when the scar tissue that naturally forms around the implant tightens and becomes unusually hard).